Publish Date (HKT) 2021-07-13
[FALSE] Does the Pfizer vaccine lead to higher rates of death and severe adverse reactions than other vaccines? Were its animal tests halted due to animal deaths?
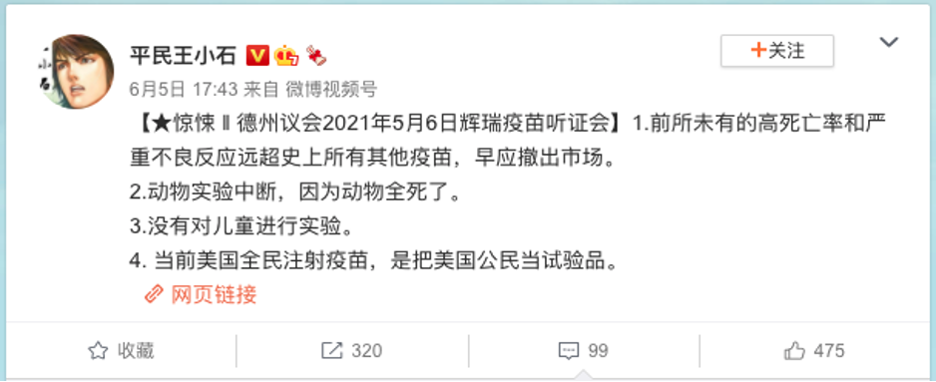
Screenshot of the Weibo post.
The Chinese-language post translates into English as:
[In a Texas Senate hearing about the Pfizer vaccine]
1. There are unprecedented severe adverse reactions to the vaccine that are much higher than other vaccinations, and we should discontinue the Pfizer vaccine as soon as possible.
2. Animal tests were halted, because the animals were all dying.
3. There have been no trials on children.
4. Because Americans are the first to be administered the vaccine, Americans are now the guinea pigs.
The Claim and Our Verdict
- The Pfizer vaccine leads to higher rates of death and severe adverse reactions than other vaccines.
- The animal trials of the Pfizer vaccine were halted, because all animals died during the tests.
- The claim originates from a public hearing held May 6, 2021, by the Texas Senate Committee on State Affairs. During the public hearing, Texas state Sen. Bob Hall, a Republican, claimed that his statement was based on information from a TV website.
- The study about the safety and effectiveness of BNT162b2 was published in the New England Journal of Medicine on Dec. 31, 2021. BNT162b2 was a COVID-19 vaccine candidate jointly developed by Pfizer and BioNTech and rolled out later as the Pfizer-BioNTech vaccine. The study concluded that after a median two-months observation period, the safety of a two-dose vaccination of BNT162b2 was similar to that of other viral vaccines. Therefore, the claim “the Pfizer vaccine has higher rates of death and severe adverse reactions than other vaccines” is false.
- The vaccination report shows that the vaccine was already successful in animal testing, specifically in non-human primates and mice, without any significant safety issues reported. There is no evidence that test animals were dying during the experiment.
News Brief
A Weibo post published June 5, 2021, claimed that the Texas Senate held a public hearing May 6, 2021, and concluded that the Pfizer vaccine has higher rates of mortality and severe adverse reactions than other vaccines. The animal trials were purportedly halted, because all the animals died during the tests.
According to its Weibo profile, the account “Civilian Wang Xiaoshi (平民王小石)” is a verified account with more than 6.45 million followers.
As of the issuance of the report, the post had been shared 320 times, and had received 99 comments and 475 likes.
Fact-checking
The Texas Senate Committee on State Affairs held a public hearing May 6, 2021. The Texas Senate discussed a bill during the hearing titled, “Relating to prohibited discrimination regarding vaccination status and mandates for receiving or participating in the administration of vaccines; authorizing administrative penalties.” The participants included Texas state senators Bob Hall (a Republican) and Eddie Lucio (a Democrat), and Dr. Angelina Farella. Hall reportedly promulgated debunked misinformation about COVID-19 and vaccines. Farella is a pediatrician with the private medical company A Brighter Tomorrow Pediatrics.

Video screenshot of Dr. Angelina Farella speaking during the public hearing.
During her testimony, Farella claimed the Pfizer vaccine’s rates of death and severe adverse reactions were much higher than other vaccines. Hall alleged the Pfizer vaccine’s animal trials were paused, because animals were dying. Hall said his claim was based on the information he had read on a TV website. HKBU FactCheck found no such TV website related to the claim.
Transcript of the public hearing (excerpt, 43:50-46:00):
Hall: With your experience, you’re talking about going back to 1999 or may before, is there been another vaccine that had the high incidence of serious hospitalizations and deaths that this vaccine is now showing?
Farella: Not, not to this extent absolutely. This one, not even close, not even close.
Hall: any other vaccines would have been pulled from the market?
Farella: Absolutely, it would have been pulled probably within the first review, just as we’ve seen in the past.
Hall: And have you seen any other vaccines that [were] put out for the public that skipped the animal test?
Farella: Never before — especially for children.
Hall: What I have read, they actually started the animal test and because the animals were dying, they stopped the test. Folks, I think that’s important to understand there. That what we’re talking about is, the American people are now the guinea pigs. This is the test program that’s going on. They didn’t do the human testing and they stopped the animal test because the animals were dying, and then they turned it out for the public. …
Lucio: Question to Senator Hall — that’s a pretty strong statement that you have data that we can look at to see what happened on those…
Hall: It’s on the TV website.
Lucio: Can you direct us to that website. I’d like to see how many they have tested that impact them to stop testing because animals were dying.
Hall: I will get that for you.
Lucio: Thank you.
Does the Pfizer vaccine lead to higher rates of death and severe adverse reactions than other vaccines?
A keyword search found an article, published in the New England Journal of Medicine on Dec. 31, 2020, about the safety and effectiveness of BNT162b2. BNT162b2 was a COVID-19 vaccine candidate jointly developed by Pfizer and BioNTech. The vaccine was later released as the Pfizer-BioNTech vaccine. The journal’s website claims it is recognized as the leading medical journal in the world. The journal has a history of more than 200 years and is ranked first in 2019 in the discipline of “Medicine, General & Internal,” based on its impact factor. The credibility of the journal, therefore, is well acknowledged.
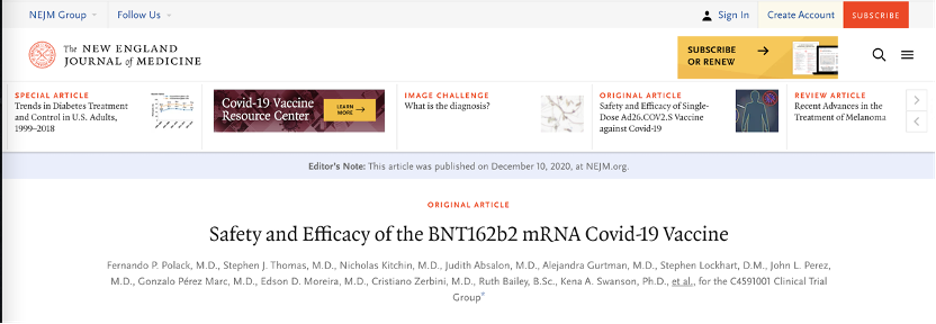
A screenshot of the article published in the New England Journal of Medicine.
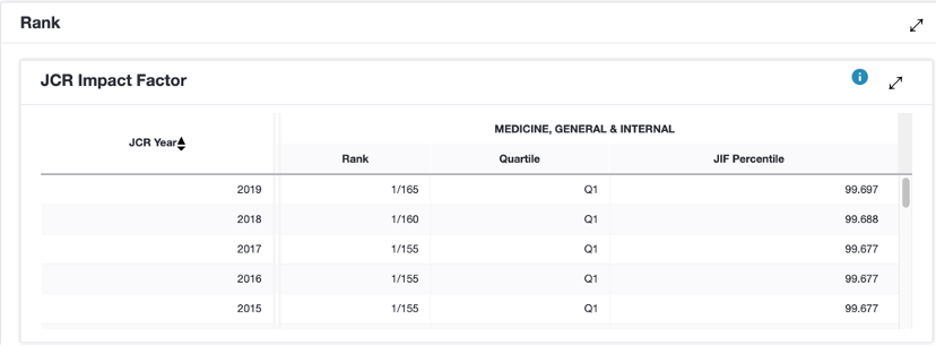
A screenshot of the impactor factor and ranking of the New England Journal of Medicine.
The study concluded, “A two-dose regimen of BNT162b2 conferred 95% protection against COVID-19 in persons 16 years of age or older. Safety over a median of 2 months was similar to that of other viral vaccines.”
Concerning side effects, the study showed that reactogenicity – a physical inflammatory response to a vaccine – was generally mild or moderate. “Only a few participants in either group had severe adverse effects, serious adverse effects, or adverse effects leading to withdrawal from the trial,” according to the study’s experimental results. A side effect of severe fatigue was about 4%, which is lower than other vaccines for the elderly. There were two cases of death in the experimental group, one of which was caused by arteriosclerosis and the other caused by cardiac arrest. There were four deaths in the control group. No deaths were considered by the investigators to be related to the vaccine or the placebo.
According to the study results above, it demonstrates a satisfactory safety rate of the candidate vaccine BNT162b2 (later approved as the Pfizer vaccine). The safety rate is similar to or lower than other viral vaccines. Therefore, the claim the Pfizer vaccine has higher rates of death and severe adverse reactions than other vaccines is FALSE.
Were the animal trials of the Pfizer vaccine halted, because all the animals died during the tests?
On Sept. 9, 2020, Pfizer and BioNTech jointly issued a press release about the pre-clinical research data of the mRNA-based vaccine candidate, BNT162b2. The press release claimed the vaccine candidate had been tested on rhesus macaques (non-human primates) and mice, and it could induce protective effects against the coronavirus. The manuscript concerning these data has been published on a preprint server of BioRxiv. It indicates that the vaccine had finished the animal trials and had demonstrated adequate protective effects.
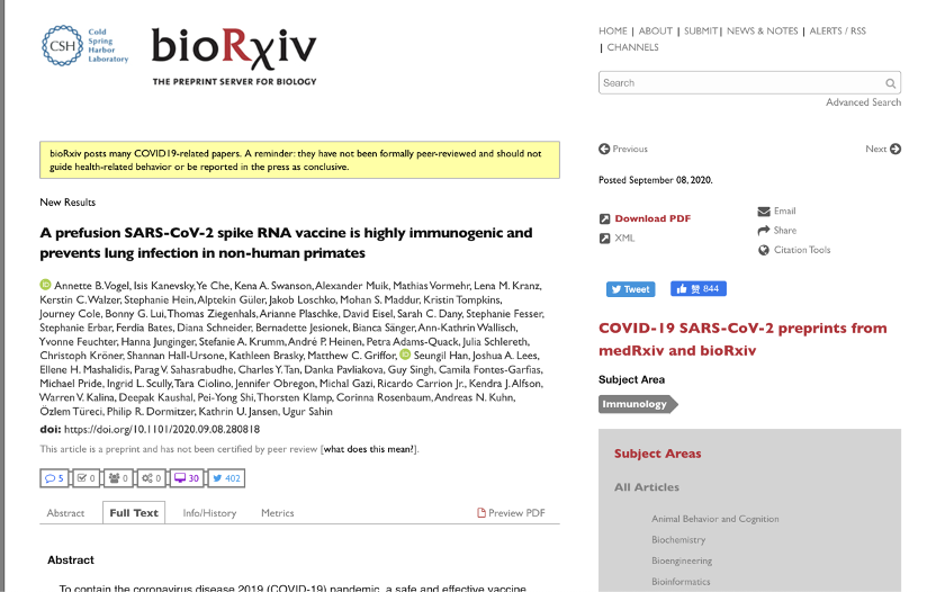
Screenshot of the preclinical data of BNT162b2 published on BioRxiv.
A peer-reviewed article was published in Nature, a multidisciplinary scientific journal, on Feb. 1, 2021. The study was about the pre-clinical trials of two COVID-19 vaccine candidates, BNT162b1 and BNT162b2. BNT162b2 was approved later as a Pfizer vaccine. The trials were conducted on rhesus macaques (Macaca mulatta). The article concluded, “BNT162b2 protects the lower respiratory tract against the presence of viral RNA and shows no evidence of disease enhancement.” It, therefore, manifests that in animal tests, the vaccine candidate BNT162b2 is safer than BNT162b1.

Screenshot of the article published in Nature.
According to the Pfizer-BioNTech COVID-19 Vaccine Emergency Use Authorization Review Memorandum issued by the US Food and Drug Administration on Dec. 11, 2020, Pfizer submitted studies regarding the vaccine’s animal tests on Wistar Han rats. “Based on nonclinical toxicity assessments, there are no significant safety issues to report,” the memorandum concluded.
The above research shows that the Pfizer vaccine has already carried out several animal tests during its pre-clinical trial stage. According to relevant reports, the vaccine showed adequate protective effects on non-human primates and did not show any significant safety issues. There is no evidence supporting the claim that the animal trials of the Pfizer vaccine were halted, because all animals died during the tests.
Conclusion
Therefore, we rate both claims as FALSE.
References
- Weibo post, June 5, 2021.
- The Weibo account “Civilian Wang Xiaoshi,” profile.
- Texas Senate State Affairs Committee, the public hearing video recording, May 6, 2021.
- The Texas Tribune, “Texas GOP state senator urged use of unproven hydroxychloroquine treatment for COVID-19. Now he’s spreading doubt about vaccines,” May 17, 2021.
- Texas Legislature Online, Legislation history, “Relating to prohibited discrimination regarding vaccination status and mandates for receiving or participating in the administration of vaccines; authorizing administrative penalties.”
- Privia Medical Group, Dr. Angelina Farella, the biography.
- Pfizer, “Pfizer and BioNTech announce data from preclinical studies of mRNA-based vaccine candidate against COVID-19,” September 9, 2020.
- The New England Journal of Medicine, about NEJM.
- BioRxiv, “A prefusion SARS-CoV-2 spike RNA vaccine is highly immunogenic and prevents lung infection in non-human primates,” September 8, 2020.
- The New England Journal of Medicine, “Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine,” December 31, 2020.
- Nature, “BNT162b vaccines protect rhesus macaques from SARS-CoV-2,” February 1, 2021.
- US Food and Drug Administration, “Pfizer-BioNTech COVID-19 Vaccine Emergency Use Authorization Review Memorandum,” November 20, 2020.












 :
: