Publish Date (HKT) 2022-06-08
A study on an mRNA-based vaccine candidate falsely attributed to the Pfizer-BioNTech vaccine

Screenshot of the Facebook post.
(*Click to view larger image)
The Claim and Our Verdict
- The claim: A Facebook post quoted a study and claims that the study proves the Pfizer-BioNTech vaccine will cause decreases in a recipient’s lymphocyte count.
- Fact-checking:
- BNT162b1 and BNT162b2 are two COVID-19 vaccine candidates jointly developed by BioNTech and Pfizer. Development of the BNT162b1 vaccine was halted in November 2020 and the BNT162b2 vaccine, marketed as Comirnaty, was listed by the World Health Organization (WHO) for emergency use on Dec. 31, 2020 and was officially approved by the U.S. Food and Drug Administration (FDA) on Aug. 23, 2021.
- The Facebook post cited a paper published in Nature. The research focus of this paper is the COVID-19 vaccine candidate BNT162b1, not the Comirnaty COVID-19 vaccine (BNT162b2). Therefore, the study does not prove that receiving the Pfizer-BioNTech vaccine will result in a decrease in the lymphocyte count in the human body.
- Our verdict: The study on an mRNA-based vaccine candidate posted on Facebook was falsely attributed to the Pfizer-BioNTech vaccine.
News Brief
A Facebook post published April 16, 2022 claims that a study proves that the Pfizer-BioNTech vaccine will cause a decrease in a recipient’s lymphocyte count. The post, written in traditional Chinese, translates as: “Will the BNT162B2 (Comirnaty) vaccine strengthen your immune system? According to a report titled ‘Phase I/II study of COVID-19 RNA vaccine BNT162b1 in adults,’ after being vaccinated with BNT162B2, the body’s lymphocytes (a type of white blood cell) will decrease.”
Lymphocytes are a type of white blood cell that is fundamentally important in the human immune system. It determines the specificity of the immune system to infectious microorganisms and other foreign substances. The post indicates that the Pfizer-BioNTech vaccine will cause decreases in the number of lymphocytes, thus, weakening the immune system.
As of the publication of this report, the post had been shared 68 times, and had received eight comments and 274 likes or reactions.
Fact-checking
The two vaccine code names, BNT162b1 (henceforth, b1) and BNT162b2 (henceforth, b2), mentioned in the Facebook post, refer to the two COVID-19 vaccine candidates jointly developed by the German-based company, BioNTech, and the U.S.-based company Pfizer. They both utilized messenger ribonucleic acid (mRNA) technology. During the vaccines’ phase one trials, the b2 vaccine was associated with a lower incidence and severity of systemic reactions than the b1 vaccine. The clinical trial of the b1 vaccine was halted in November 2020. The World Health Organization (WHO) listed the b2 vaccine, branded as Comirnaty, for emergency use on Dec. 31, 2020. The U.S. Food and Drug Administration (FDA) authorized the Comirnaty COVID-19 vaccine (b2 vaccine) for emergency use on Dec. 11, 2020, and approve the vaccine on Aug. 23, 2021 for the prevention of COVID-19 in individuals 16 years of age and older. Under the Prevention and Control of Disease (Use of Vaccines) Regulation (Cap. 599K), Hong Kong’s health authority authorized the Comirnaty COVID-19 vaccine on Jan. 25, 2021 for emergency use in Hong Kong.
The Facebook post cited a paper published Aug. 12, 2020 in Nature, titled “Phase I/II study of COVID-19 RNA vaccine BNT162b1 in adults.” The research focus of this paper is the b1 vaccine, not the b2 vaccine which has been rolled out and marketed as Comirnaty. The Facebook post confused the b1 vaccine with the b2 vaccine, and falsely attributed the study on another COVID-19 vaccine candidate to the Pfizer-BioNTech vaccine. Therefore, the claim that “this study proves the Pfizer-BioNTech vaccine will cause decreases in lymphocyte count” is false.
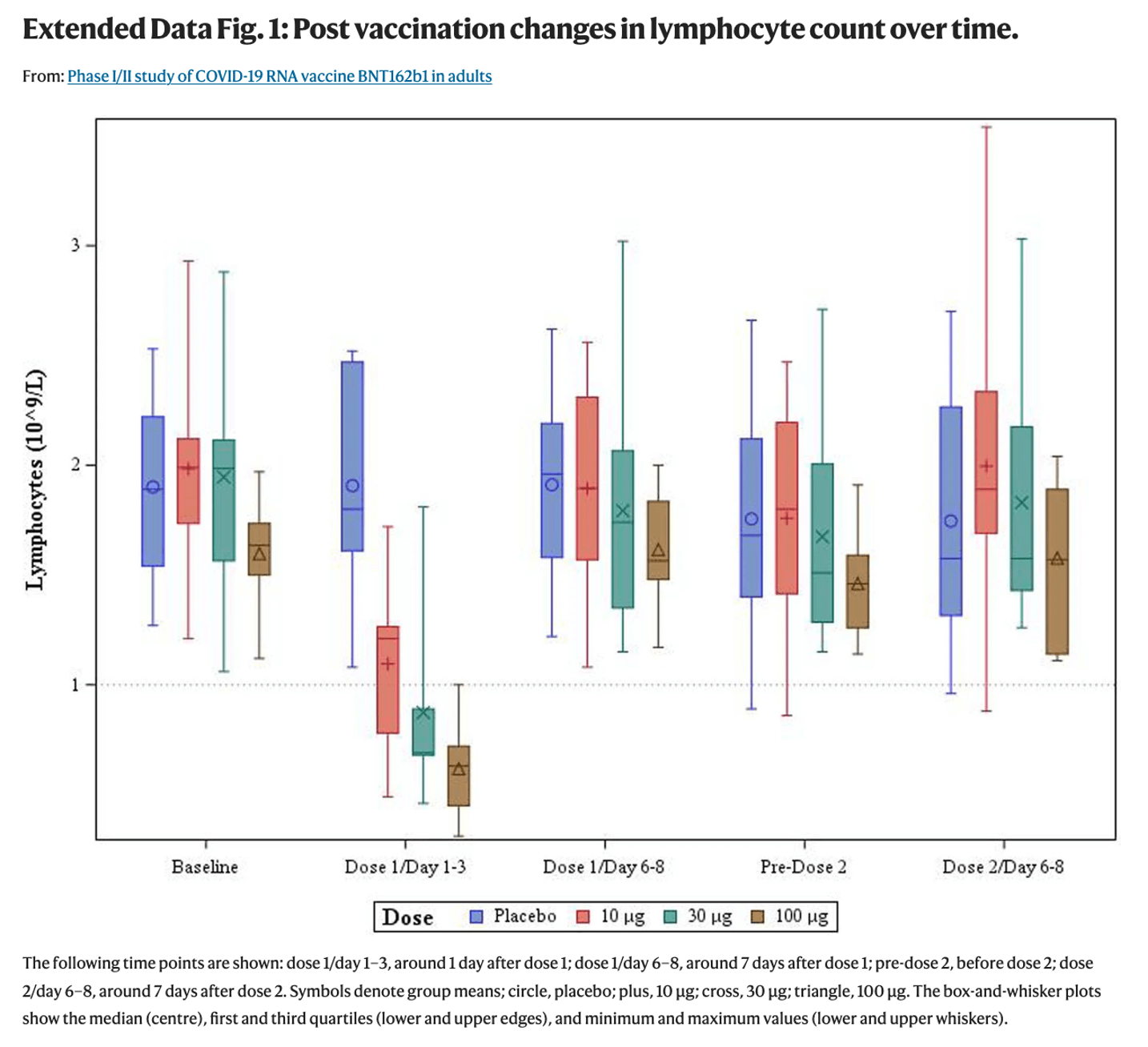
Moreover, although some participants in the study experienced a decrease in lymphocyte counts after receiving the b1 vaccine, the paper concluded that the decrease was transient: some participants experienced a decrease in lymphocyte counts about one day after vaccination, but the lymphocyte counts returned to normal six to eight days after being vaccinated.
Post-vaccination changes in lymphocyte count over time.
(*Click to view larger image)
Conclusion
The study on an mRNA-based vaccine candidate posted on Facebook was falsely attributed to the Pfizer-BioNTech vaccine.
References
- Facebook, post, April 16, 2022.
- The New England Journal of Medicine, “Safety and Immunogenicity of Two RNA-Based Covid-19 Vaccine Candidates,” Dec. 17, 2020.
- Reuters, “China’s Fosun to end BioNTech’s COVID-19 vaccine trial, seek approval for another,” Nov. 4, 2020.
- S. Food and Drug Administration, “Comirnaty and Pfizer-BioNTech COVID-19 Vaccine.”
- Hong Kong Electronic Legislation, “Prevention and Control of Disease (Use of Vaccines) Regulation.”
- Hong Kong Government, “Secretary for Food and Health authorises COVID-19 vaccine by Fosun Pharma/BioNTech for emergency use in Hong Kong,” Jan. 25, 2021.
- Food and Health Bureau, “Authorisation of COVID-19 Vaccines under the Prevention and Control of Disease (Use of Vaccines) Regulation (Cap.599K).”
- WHO, “WHO issues its first emergency use validation for a COVID-19 vaccine and emphasizes the need for equitable global access,” Dec. 31, 2020.
- Nature, “Phase I/II study of COVID-19 RNA vaccine BNT162b1 in adults,” Aug. 12, 2020.












 :
: